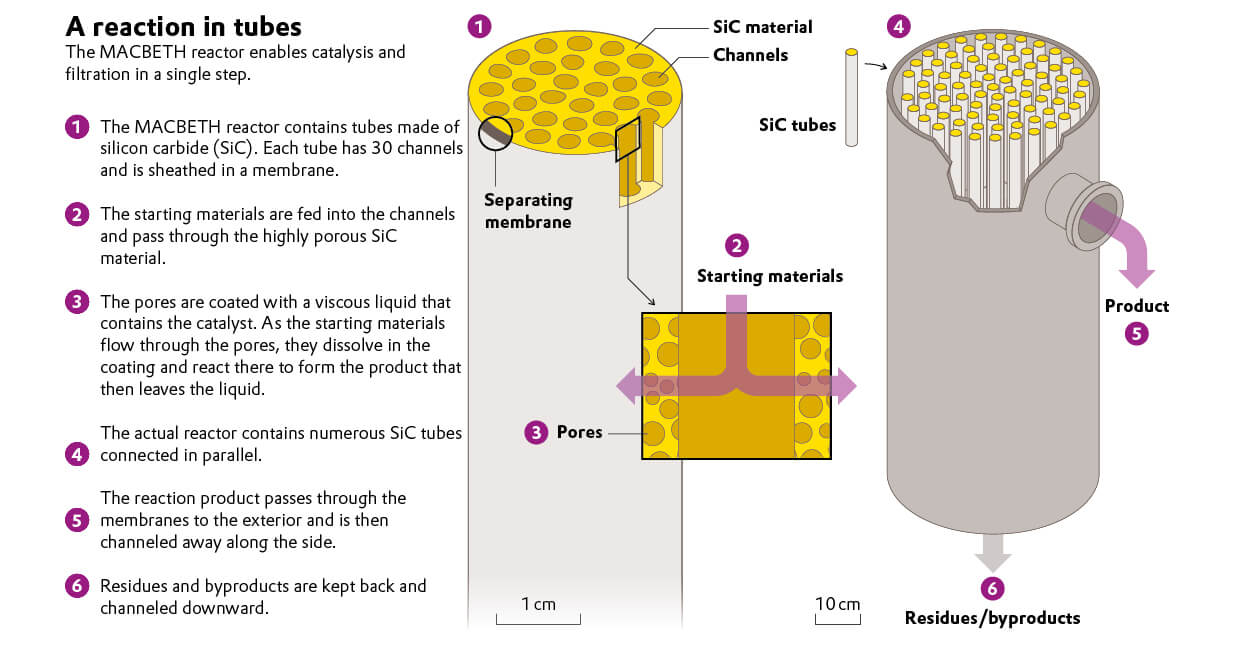
In hydroformylation processes, catalysts are normally dissolved in the reactor liquid. Known as homogeneous catalysis, this process is very efficient, but also has its drawbacks. “The catalyst mixes with the product and then has to be separated with great effort,” explains Robert Franke, the head of hydroformylation research at Evonik. Moreover, it breaks down over time and has to be reprocessed. Although a solid catalyst prevents such mixing, the yield is generally much lower because the reactants and the catalyst do not come into contact as closely. In the ROMEO project, researchers looked for a way out of this dilemma. Their idea was to take advantage of the fact that very porous materials have a large surface area. What would happen if the surface of the pores was coated with the catalyst, in the form of a viscous film, for example? Franke had read about researchers from Denmark and Erlangen who dissolved catalysts in ionic liquids that were used to coat the pores. Ionic liquids are salts that liquefy at temperatures of about 100 degrees Celsius. Experts in this field also took part in the ROMEO project, as did the Danish company LiqTech and its very porous ceramic tubes. In addition, the outer wall of the tubes was coated with a membrane that only lets the reaction product of the hydroformylation pass through. This is not a trivial matter. For example, the aldehyde molecules that are produced during hydroformylation are larger than the starting materials that remain in small amounts in the gas flow. During the ROMEO project, the researchers found a siloxane polymer to be the right material for the membrane. It holds back the starting materials while the aldehyde molecules dissolve in the polymer. Once they have dissolved in the membrane, the molecules travel to the outside, where they are released and can be channeled away.