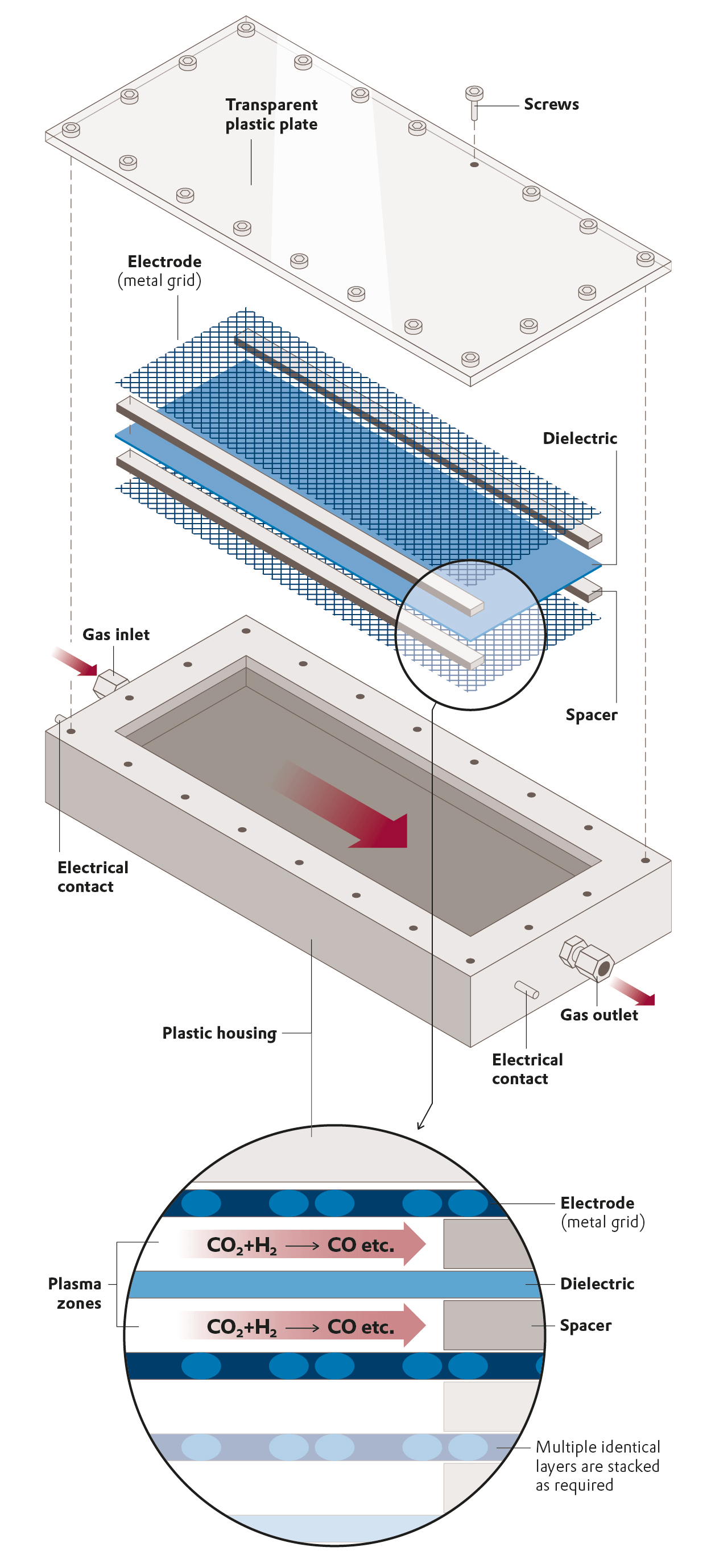
Efficiency enhancement
Microreactors from the 3D printer
Additive manufacturing opens the door to lower CO2 emissions and less energy consumption in chemical production.

The circular economy
Used catalysts for the energy transition
A unique technology from Evonik enables old catalysts to be rejuvenated. This cuts greenhouse gas emissions.

Chemical production
The Direct Route
Evonik plans to use a new method to make the production of propylene glycol more sustainable.

Plastic
Turning emissions into raw materials
Evonik researchers are working to use emissions of the steel industry for the production of plastic.